Solvent Recovery in Extraction Process: How It Works and Why It Matters
In almost every modern extraction process — from plant extracts and pharmaceuticals to paints and specialty chemicals — solvents are the invisible workhorses. They dissolve target compounds, carry them through filters and columns, and then are usually evaporated or distilled away. But buying large volumes of solvent, managing hazardous waste and complying with environmental rules can become a serious burden.
This is where solvent recovery in extraction process becomes a practical, high‑impact solution. By recovering and reusing solvents with a dedicated solvent recycler machine, extraction lines can reduce operating costs, cut hazardous waste and improve sustainability, often with a payback measured in months rather than years.
What Is Solvent Recovery in Extraction Process?
In simple terms, solvent recovery means separating used solvent from contaminants and bringing it back to a usable purity level, instead of discarding it as waste. In extraction, this usually happens after solvents have:
Dissolved active compounds (for example, cannabinoids, herbal actives, flavors or fragrances)
Picked up oils, pigments, resins or fines from the raw material
Been mixed or diluted with process water or other chemicals
Most recovery systems rely on distillation: the used solvent is heated so it evaporates, vapors are condensed back to liquid, and the heavier contaminants remain in a residue tank. Properly designed systems can consistently achieve around 95% recovery of the solvent charge.
Why Solvent Recovery Is Critical for Extraction Operations
For extraction facilities, the decision to install solvent recycling equipment is rarely just about “being green”. It usually starts with three very practical drivers.
1. Direct cost savings on solvent purchase
Solvents such as ethanol, isopropanol and acetone are not cheap, especially at high purity. When 70–90% of each batch is normally recoverable, discarding used solvent means throwing away money. A dedicated solvent recovery unit allows that solvent to be reused many times before final disposal is necessary.
2. Lower hazardous waste volumes
Used solvent mixed with oils, pigments and other organics is usually classified as hazardous waste. Disposal costs per liter can be several times the purchase price of fresh solvent. By distilling off clean solvent and only discarding the concentrated residue, total waste volume drops dramatically.
According to a 2022 study by the U.S. Environmental Protection Agency on solvent‑intensive manufacturing, on‑site recovery can reduce solvent waste volumes by 60–95% compared with one‑time use and off‑site disposal, depending on process stability and solvent type.
3. Compliance and sustainability
Regulators worldwide increasingly expect companies to minimize volatile organic compound (VOC) emissions and hazardous waste. Solvent recovery fits directly into this trend:
Less waste shipped off‑site
Lower emissions from solvent handling
Stronger sustainability story for customers and investors

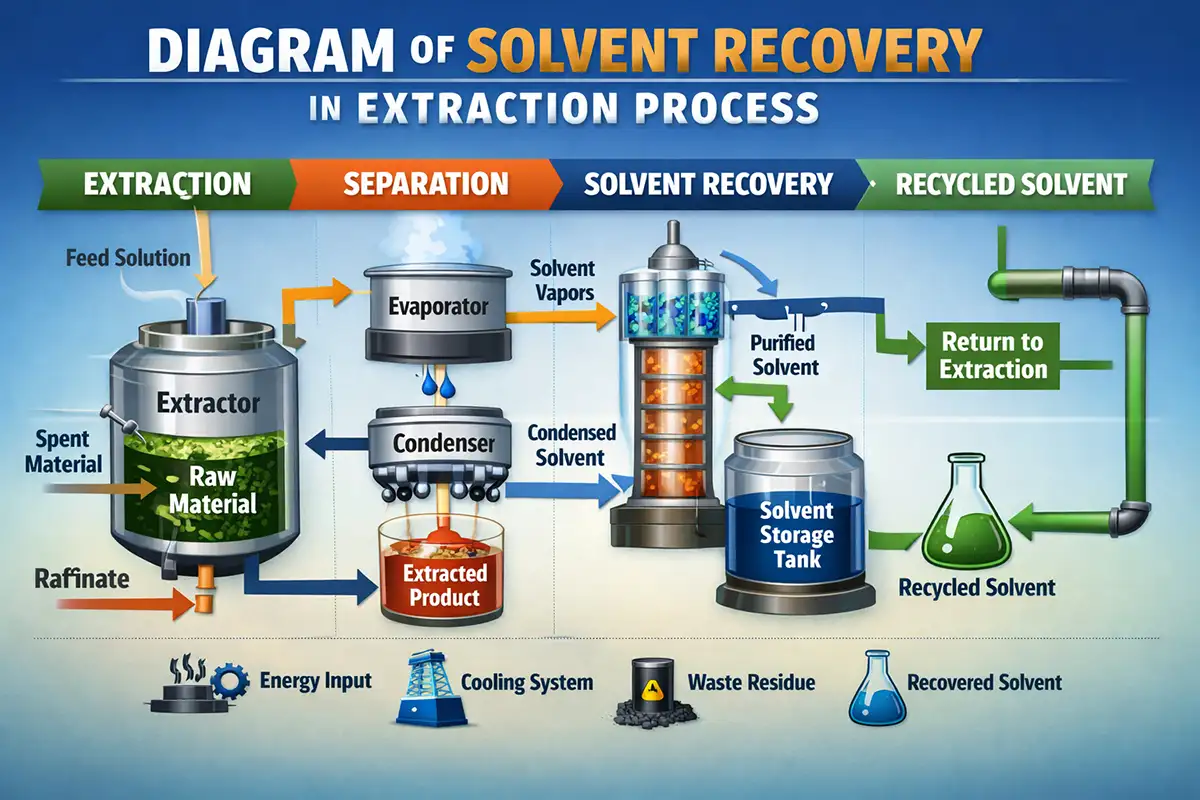
How Solvent Recovery Works in an Extraction Line
While every plant is different, most extraction processes that use solvent recovery follow a similar pattern:
Extraction: Fresh solvent dissolves target components from raw material.
Separation: Solid and liquid phases are separated (filtration, centrifugation, etc.).
Primary solvent removal: The bulk of the solvent is evaporated or distilled to concentrate the extract.
Used solvent collection: The “dirty” solvent is collected in a feed tank.
Solvent recovery: A solvent recycler machine distills the used solvent, yielding clean distillate and a concentrated residue.
Solvent reuse: Recovered solvent returns to the extraction process, often after a quick quality check.
A common question at this stage is whether the recovered solvent will have the same performance as fresh solvent in terms of extraction yield and selectivity.
Key Features of a Modern Solvent Recycler Machine
A modern solvent recovery machine is more than a simple boiler and condenser. It is engineered for safe, repeatable and efficient operation in industrial environments, especially for flammable solvents.
Below is an example set of explosion‑proof solvent recyclers suitable for extraction operations, with capacities from 20 L to 400 L per batch:
| Model | Feed Capacity (L) | Heating Power (kW) | Treatment Time (min) | Recovery (%) | Machine Weight (kg) | Machine Size (mm) |
|---|---|---|---|---|---|---|
| T-20Ex | 20 | 2 | 120 | 95 | 153 | 860 × 760 × 1190 |
| T-60Ex | 60 | 4 | 150 | 95 | 170 | 1160 × 870 × 1260 |
| T-80Ex | 80 | 5 | 180 | 95 | 200 | 1180 × 850 × 1290 |
| T-125Ex | 125 | 6 | 210 | 95 | 280 | 1250 × 920 × 1450 |
| T-250Ex | 250 | 16 | 240 | 95 | 520 | 2600 × 1200 × 1950 |
| T-400Ex | 400 | 32 | 270 | 95 | 1200 | 1990 × 1850 × 2090 |
For more detail on commercial configurations, the product pages for asolvent recycling systemor asolvent recycling machineshow how these specifications translate into complete turnkey solutions.
Core design elements to focus on
Explosion‑proof design (Ex): Essential for flammable solvents such as ethanol or isopropanol, including certified electrical components and proper grounding.
Temperature control: A range up to 200 °C allows processing of common organic solvents without thermal degradation.
Recovery rate: Around 95% recovery minimizes solvent loss and speeds up payback.
Batch size: From 20 L laboratory or pilot units to 400 L industrial units, the right batch volume must match actual solvent generation per shift.
Ease of residue discharge: Efficient residue handling reduces downtime and exposure to concentrated waste.

How to Size a Solvent Recovery System for an Extraction Process
Sizing is where many projects either succeed or struggle. Oversized machines tie up capital; undersized units become bottlenecks. There is a straightforward way to approach this.
1. Quantify daily used‑solvent volume
Start with the total volume of solvent leaving the extraction process as “dirty” solvent per shift or per day. For example:
Extraction line uses 1000 L of ethanol per 8‑hour shift.
80% of that ends up as recoverable dirty solvent.
Daily recovery target ≈ 800 L.
2. Match batch size and cycle time
Assume a treatment time of 180–240 minutes for a mid‑range unit under typical contamination. A 250 L machine (T‑250Ex) can therefore realistically process two to three full batches per shift, or roughly 500–750 L per 8‑hour period.
In the example above, either:
Use one 400 L unit (T‑400Ex) to handle the full 800 L per shift, or
Use two 250 L units (2 × T‑250Ex) for redundancy and scheduling flexibility.
3. Consider growth and maintenance
It is normally wise to allow 20–30% capacity margin for:
Future production increase
Downtime during cleaning or maintenance
Unexpectedly dirty batches requiring longer cycles
Safety and Best Practices in Solvent Recovery
Because many extraction solvents are flammable or toxic, safety is non‑negotiable when planning solvent recovery in extraction process steps.
Core safety points
Explosion‑proof classification: Ensure the unit is suitable for the solvent type and local regulations.
Ventilation: Adequate airflow and vapor management around the solvent recycler.
Temperature limits: Stay within the machine’s specified range (typically up to 200 °C) and well below solvent decomposition temperatures.
Operator training: Clear procedures for loading, unloading, cleaning and emergency response.
Facilities that already deal with solvent waste can benefit from general waste‑handling guidance such as the article on how to dispose of solvent waste, which explains broader safety and regulatory considerations.
When Does Solvent Recovery Make Sense?
In practice, solvent recovery is most attractive when:
Solvent cost per liter is high.
Solvent consumption is steady or increasing.
Hazardous waste disposal costs are significant.
Environmental permits or audits demand better waste reduction.
For small, irregular laboratory operations, simple bench‑top distillation might be enough. But once daily dirty‑solvent volumes reach tens of liters or more, a dedicated solvent recovery machine quickly becomes more efficient, safer and easier to operate.
Turn Solvent from a Cost Center into a Circular Resource
Solvent recovery in extraction process is one of the most direct ways to reduce costs, support compliance and improve sustainability — without changing core formulas or products. With recovery rates around 95% and capacities from 20 L to 400 L per batch, a properly selected solvent recycler can usually pay for itself in a short time.
